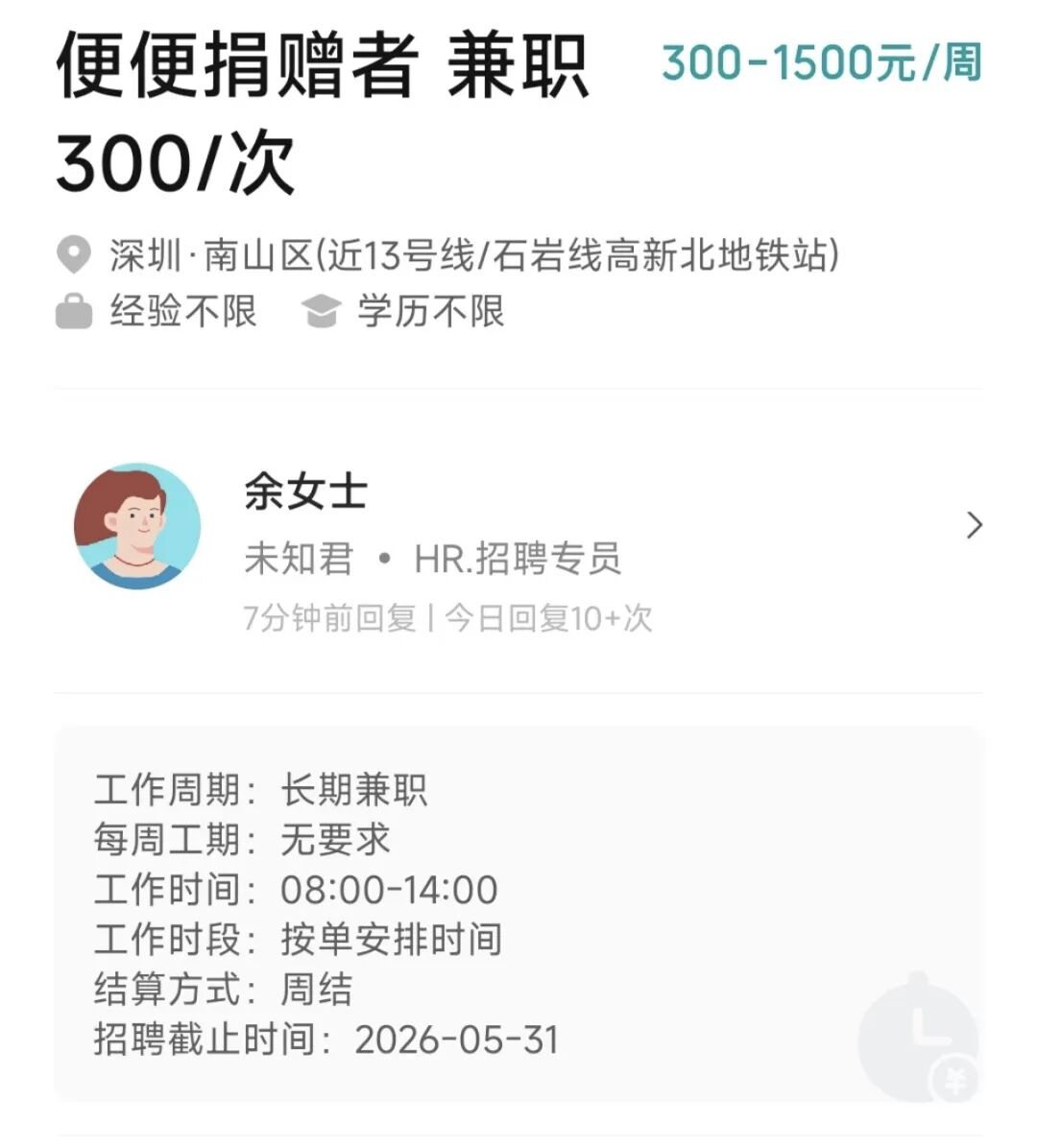
捐屎一次 3000 元,一年能赚 120 万?他们向 5 万人买了 4 吨大便

专属客服号

微信订阅号
大数据治理
全面提升数据价值
赋能业务提质增效



监管层面也随之跟进。
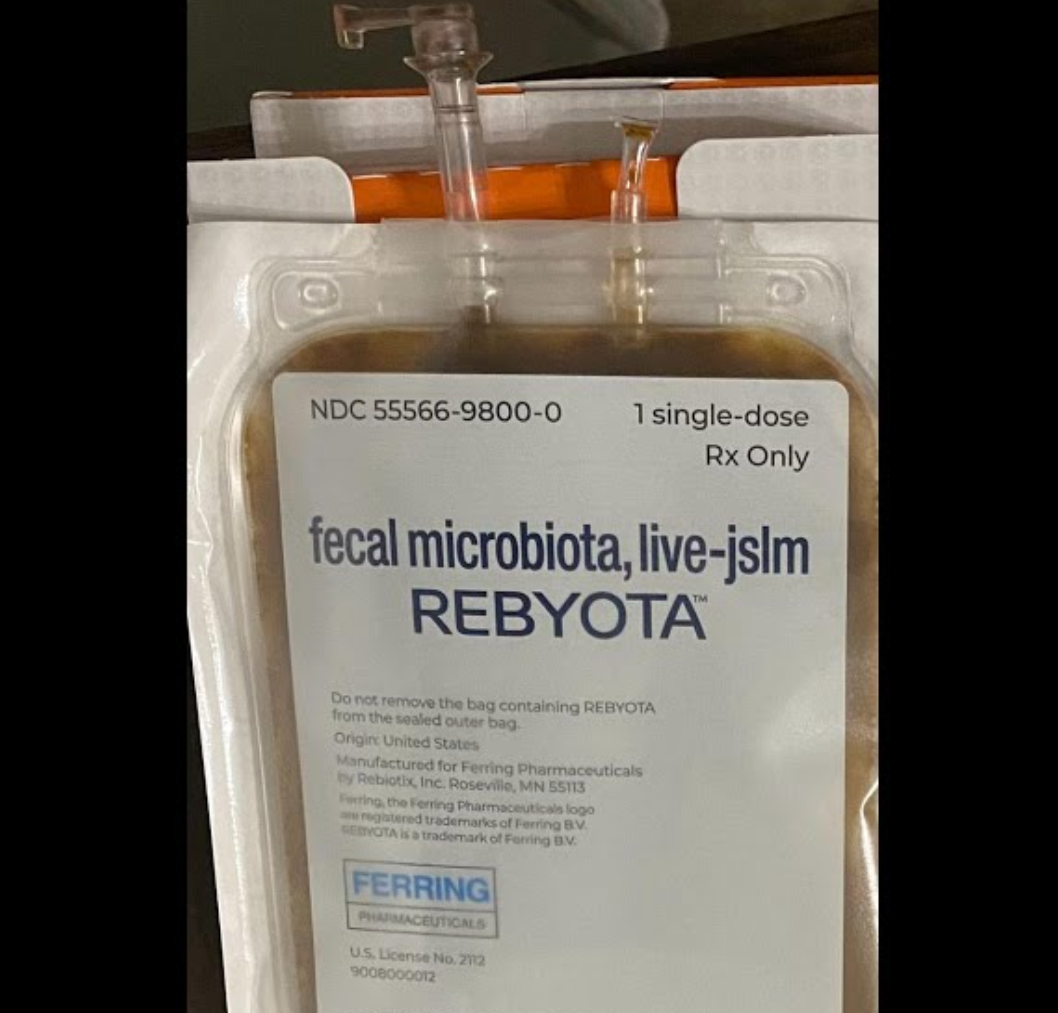
2024 年,美国胃肠病学会(AGA)发布了首部专门针对 FMT 及菌群疗法的临床实践指南,对传统 FMT 与两款获批产品在艰难梭菌感染、炎症性肠病、肠易激综合征中的使用场景分别作出明确推荐——标志着这一领域从「实验性操作」正式进入规范化临床路径。
而在中国,「粪便移植」也正在从默许走向规范。
2025 年 8 月,国家医保局印发《消化系统医疗服务价格项目立项指南(试行)》,正式设立「肠道菌群移植费」收费项目——这意味着 FMT 在中国医疗体系内取得了合法身份。

国家医保局文件截图
策划:z_popeye|监制:islay
参考文献:
[1]Dubberke E R, Han Z, Bobo L, et al. Impact of clinical symptoms on interpretation of diagnostic assays for Clostridium difficile infections[J]. Journal of clinical microbiology, 2011, 49(8): 2887-2893.
[2]Van Nood E, Vrieze A, Nieuwdorp M, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile[J]. New England Journal of Medicine, 2013, 368(5): 407-415.
[3]https://www.statnews.com/2020/12/08/open-biome-lost-year/
[4]https://www.statnews.com/2020/03/12/two-dead-four-hospitalized-stool-transplant/
[5]https://www.fda.gov/safety/medical-product-safety-information/fecal-microbiota-transplantation-safety-alert-risk-serious-adverse-events-likely-due-transmission
[6] https://www.forbes.com/profile/mark-smith-1/?sh=249bbe362b2f
[7]https://paw.princeton.edu/article/tigers-week-nonprofit-entrepreneurs-mark-smith-09-and-james-burgess-09
[8]EISEMAN B, SILEN W, BASCOM GS, et al. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery. 1958;44(5):854-9.
[9]van Nood E, Vrieze A, Nieuwdorp M, et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368(5):407-15. doi: 10.1056/NEJMoa1205037
[10]https://www.biomebank.com/stool-donation/
[11]https://www.fda.gov/safety/medical-product-safety-information/fecal-microbiota-transplantation-safety-alert-risk-serious-adverse-events-likely-due-transmission
[12]DeFilipp Z, Bloom PP, Torres Soto M, et al. Drug-Resistant E. coli Bacteremia Transmitted by Fecal Microbiota Transplant. N Engl J Med. 2019;381(21):2043-2050. doi: 10.1056/NEJMoa1910437'
[13]Lodise T, Guo A, Yang M, et al. Budget Impact Analysis of REBYOTA™ (Fecal Microbiota, Live-jslm [FMBL]) for Preventing Recurrent Clostridioides difficile Infection in the US. Adv Ther. 2023;40(6):2801-2819. doi: 10.1007/s12325-023-02506-0
[14]https://www.biose.com/food-for-thought-2024/
[15]https://www.fda.gov/regulatory-information/search-fda-guidance-documents/enforcement-policy-regarding-investigational-new-drug-requirements-use-fecal-microbiota
[16]https://www.abc.net.au/news/2020-03-09/first-public-poo-bank-in-adelaide/12034138
[17]https://goodnatureprogram.com/faq/

